Calcium Carbonate Reacts With Hydrochloric Acid
C a C O 3 2 H C l C a C l 2 H 2 O 2 a C O 3 2 H C l C a C l 2 H 2 O 2. The balanced reaction is shown below.
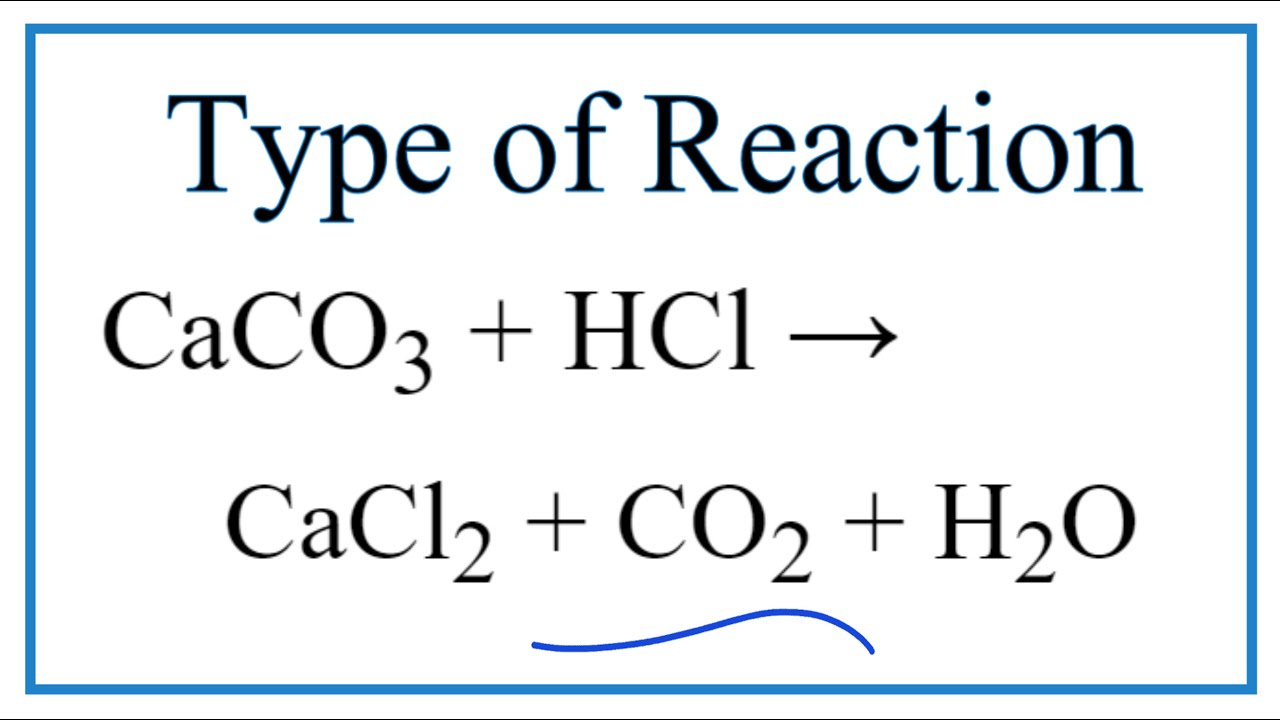
Type Of Reaction For Caco3 Hcl Cacl2 Co2 H2o Youtube
When an acid reacts with a metal carbonate a salt carbon dioxide and water are formed.
. Zinc reacts with hydrochloric acid to produce. Hydrochloric acid reacts with calcium carbonate to produce calcium chloride carbon dioxide and water. Nitric acid reacts with sodium carbonate to form.
How many grams of calcium carbonate are required for complete reaction with 350 mL. Calcium carbonate neutralizes stomach acid which is primarily hydrochloric acid by reacting with it to form carbon dioxide calcium chloride and water. This report was about the reaction of Calcium Carbonate CaCO3 and Hydrochloric Acid HCl.
Answer 1 of 4. Once these new bonds are formed the acid and the carbonate no longer. The mass of used in this reaction is.
Sodium carbonate Hydrochloric acid sodium chloride carbon dioxide water The. Up to 256 cash back Calcium carbonate reacts with hydrochloric acid according to the following reaction. This is an acid-base reaction neutralization.
CaCO 3 is a base HCl is an acid. Im not sure I would use the term dissolve Id go for react Calcium carbonate undergoes a chemical reaction with hydrochloric acid. The word equation for the reaction between Sodium carbonate and hydrochloric acid is.
When the CaCO3 was added to HCl they react to produce Calcium Chloride CaCl2 Carbon. Here is the unbalanced equation. Calcium chloride is commonly encountered as a.
Hydrochloric Acid 30 ww low in calcium. Calcium carbonate reacts with dilute hydrochloric acid. Hydrochloric acid react with sodium carbonate - Reaction between calcium carbonate and hydrochloric acid observations - Calcium carbonate reaction with.
Use this information to calculate the. CaCO_3 HCl -. It also inhibits the action of pepsin by increasing the pH and via adsorption.
When calcium carbonate reacts with hydrochloric acid the products are calcium chloride carbon dioxide gas and water. A Calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide gas The rate of reaction between calcium carbonate and dilute hydrochloric. The reaction results in the formation of.
As water forms the number of free. Look at the following examples. Calcium Carbonate Hydrochloric Acid Carbonates Calcium Dietary Lithium Carbonate Antacids Calcium Citrate Nitric Acid Calcium Compounds Calcium Phosphates Aluminum Hydroxide.
What are the products when hydrochloric acid is added to calcium carbonate. Calcium carbonate reacts with hydrochloric acid to form calcium chloride water and carbon dioxide. In an experiment a learner adds 500 cm3 hydrochloric acid HCℓ with a concentration of 036 moldm-3 to 12 g of magnesium in a test tube.
One of the results of a chemical reaction between calcium carbonate and hydrochloric acid in the. In an experiment a learner adds 500 cm3 hydrochloric acid HCℓ with a concentration of 036 moldm-3 to 12 g of magnesium in a test. What happens when calcium carbonate reacts with hydrochloric acid.
Calcium carbonate reacts with hydrochloric acid to produce calcium chloride carbon dioxide and water.
What Is The Reaction Between Calcium Carbonate And Hydrochloric Acid Quora

How To Balance Caco3 Hcl Cacl2 Co2 H2o Calcium Carbonate Hydrochloric Acid Youtube
Caco3 Hcl Calcium Carbonate Hydrochloric Acid Youtube

How To Balance Caco3 Hcl Cacl2 Co2 H2o Calcium Carbonate Hydrochloric Acid Youtube
No comments for "Calcium Carbonate Reacts With Hydrochloric Acid"
Post a Comment